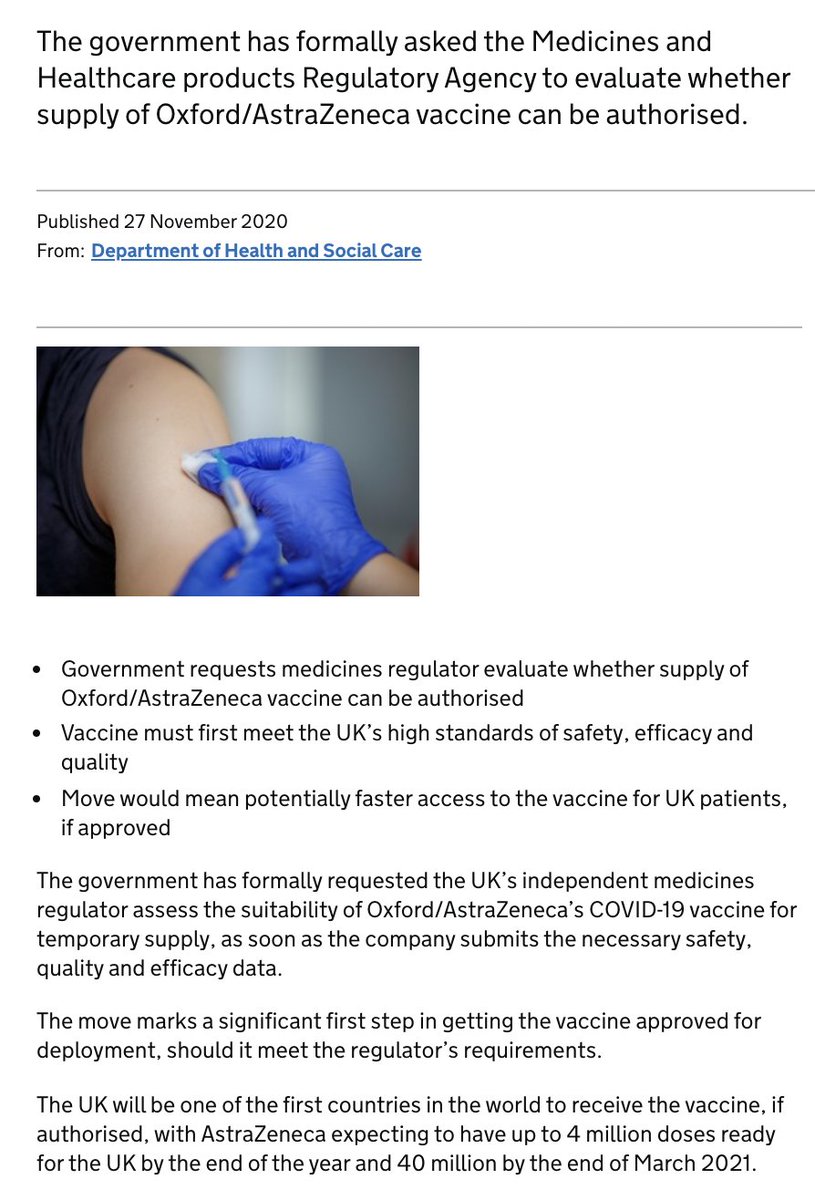
My understanding of AstraZeneca's EU doses:
- contract signed in August
- EU expected over 100 mln doses delivered by end-March, possibly 120 mln
- in early Dec, AZ revised down to 80 mln
- on Jan 22, AZ revised to 31 mln
- distributed proportionally: eg Ireland's share is ~1.1%
- contract signed in August
- EU expected over 100 mln doses delivered by end-March, possibly 120 mln
- in early Dec, AZ revised down to 80 mln
- on Jan 22, AZ revised to 31 mln
- distributed proportionally: eg Ireland's share is ~1.1%
Meanwhile, on Jan 13, AstraZeneca chief Tom Keith-Roach told a UK parliament hearing it was scaling up vaccine deliveries "very rapidly" and by mid-Feb would be able to deliver two million doses to the UK every week.
Up to that point, 1.1 mln doses had been delivered to UK.
Up to that point, 1.1 mln doses had been delivered to UK.
AZ and UK govt have represented UK doses as made in UK, and visa versa for EU.
However, on December 8, the UK's Vaccine Taskforce manufacturing lead Ian McCubbin told reporters that
"The initial supply... actually comes from the Netherlands and Germany." https://www.reuters.com/article/uk-health-coronavirus-britain-astrazenec/uks-initial-astrazeneca-shots-will-come-from-europe-taskforce-says-idUKKBN28I1NH
However, on December 8, the UK's Vaccine Taskforce manufacturing lead Ian McCubbin told reporters that
"The initial supply... actually comes from the Netherlands and Germany." https://www.reuters.com/article/uk-health-coronavirus-britain-astrazenec/uks-initial-astrazeneca-shots-will-come-from-europe-taskforce-says-idUKKBN28I1NH
The EU's understanding of its contract is: it allocated €336 million in funding to AstraZeneca, and the deal was that the company would pre-produce doses, to be ready as soon as approval was granted (probably tomorrow).
The contract specified four factories, two in UK, two in EU
The contract specified four factories, two in UK, two in EU
Dec 23: AstraZeneca applies for approval in the UK
Dec 30: AstraZeneca vaccine granted emergency approval in UK
Jan 12: AstraZeneca applies for approval in EU
Expected Jan 29: European Medicines Agency decision on conditional marketing approval (normal route, not emergency)
Dec 30: AstraZeneca vaccine granted emergency approval in UK
Jan 12: AstraZeneca applies for approval in EU
Expected Jan 29: European Medicines Agency decision on conditional marketing approval (normal route, not emergency)
Contracts:
UK signed contract with AZ in May for 100 million doses. In November, govt says 40 mln expected by end-March; company in Dec says merely "millions".
EU signed contract with AZ for 300 mln doses, with option for extra 100 mln. Until Friday, expected 80 mln by end-March.
UK signed contract with AZ in May for 100 million doses. In November, govt says 40 mln expected by end-March; company in Dec says merely "millions".
EU signed contract with AZ for 300 mln doses, with option for extra 100 mln. Until Friday, expected 80 mln by end-March.
An old lesson still not learned by UK government and others: Brussels can hear what you say publicly in Britain.
Sad background to all this:
The Oxford researchers who developed the vaccine said their know-how would be granted in "non-exclusive, royalty-free licences" during the pandemic.
Ultimately an exclusive contract with AZ was signed https://innovation.ox.ac.uk/technologies-available/technology-licensing/expedited-access-covid-19-related-ip/ https://www.astrazeneca.com/media-centre/press-releases/2020/astrazeneca-and-oxford-university-announce-landmark-agreement-for-covid-19-vaccine.html
The Oxford researchers who developed the vaccine said their know-how would be granted in "non-exclusive, royalty-free licences" during the pandemic.
Ultimately an exclusive contract with AZ was signed https://innovation.ox.ac.uk/technologies-available/technology-licensing/expedited-access-covid-19-related-ip/ https://www.astrazeneca.com/media-centre/press-releases/2020/astrazeneca-and-oxford-university-announce-landmark-agreement-for-covid-19-vaccine.html
South Africa and India led a call supported by human rights groups and @MSF for patents on Covid-19 medicines to be waived for the duration of the pandemic to allow for the widest possible production. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)32581-2/fulltext
Disparity in vaccines as of last week according to @WHO:
Rich countries: 39 million doses
Poor countries: 25 doses (Russia's Sputnik vaccine, given in Guinea)
WHO has called on rich countries not to hoard vaccines, as the pandemic will drag on longer for all without sharing doses
Rich countries: 39 million doses
Poor countries: 25 doses (Russia's Sputnik vaccine, given in Guinea)
WHO has called on rich countries not to hoard vaccines, as the pandemic will drag on longer for all without sharing doses
Key information is obscured by the fact the contract is confidential and we have only rival claims by AZ and EU about it.
EU spokesman today: "We have written to the company informing them of our intention of publishing the contract and this is now under discussion with them."
EU spokesman today: "We have written to the company informing them of our intention of publishing the contract and this is now under discussion with them."
"Production capacity" was "one of the selection criteria" for what vaccines the EU helped to fund, in exchange for early delivery agreements, according to EU spokesman.
"Therefore plants are listed" in the contract: 2 in UK, 2 in EU.
"Therefore plants are listed" in the contract: 2 in UK, 2 in EU.
EMA is expected to decide on AstraZeneca tomorrow, would be a surprise if it wasn't authorised. A relatively small number of older people took part in its trial. EMA hasn't ruled out authorising just for younger groups but we won't know until they announce https://twitter.com/Marieke83473064/status/1354765315641499649?s=20
There's the contract now, lawyers can have a look https://ec.europa.eu/commission/presscorner/detail/en/ip_21_302

 Read on Twitter
Read on Twitter