History lesson:
When anti-retrovirals were developed for HIV in the 90s, drug-makers had no interest in Africa ("not a lucrative market") & ART was unaffordable.
Zidovudine was marketed at a price of US$8000 per year!
When anti-retrovirals were developed for HIV in the 90s, drug-makers had no interest in Africa ("not a lucrative market") & ART was unaffordable.
Zidovudine was marketed at a price of US$8000 per year!
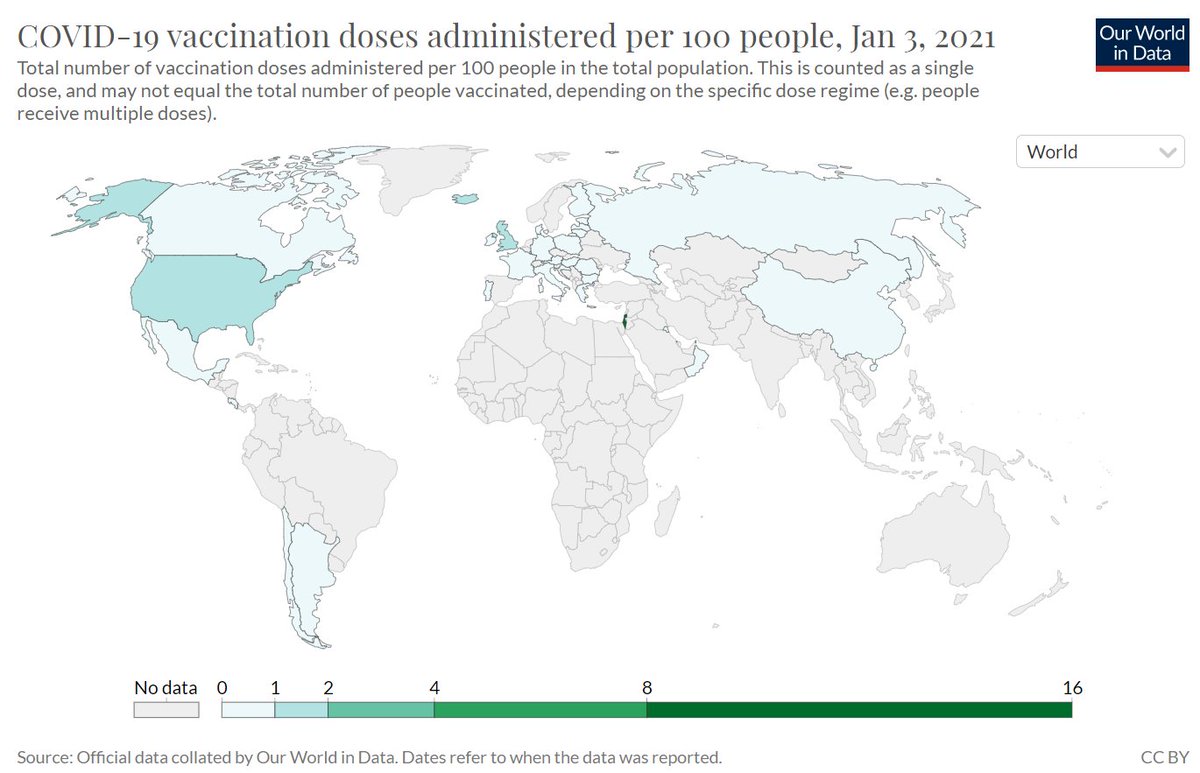
We are seeing the same lack of pharma interest in providing affordable #COVID19 vaccines for Africa https://www.bloomberg.com/news/articles/2021-01-03/pfizer-to-supply-african-health-care-workers-with-vaccines?sref=d39KtWbu
After a lot of global advocacy, widespread access to affordable ARVs became feasible after Cipla, a generics Indian manufacturer offered triple therapy for less than a dollar a day.
Full story here: …https://globalizationandhealth.biomedcentral.com/articles/10.1186/1744-8603-7-33
Full story here: …https://globalizationandhealth.biomedcentral.com/articles/10.1186/1744-8603-7-33
We need the same type of advocacy now for Covid-19 vaccine access for LMICs!
And we need generic companies in India & China to make vaccines that are effective & affordable.
We cannot wait for "trickle down" from HICs. That could take years! https://www.forbes.com/sites/madhukarpai/2020/02/17/global-health-technologies-time-to-re-think-the-trickle-down-model/?sh=11b1143344d9
And we need generic companies in India & China to make vaccines that are effective & affordable.
We cannot wait for "trickle down" from HICs. That could take years! https://www.forbes.com/sites/madhukarpai/2020/02/17/global-health-technologies-time-to-re-think-the-trickle-down-model/?sh=11b1143344d9
This is feasible today, since both India & China have approved Covid-19 vaccines.
India has provided emergency use authorization for two vaccines (Covishield & Covaxin), while has approved Sinopharm
India has provided emergency use authorization for two vaccines (Covishield & Covaxin), while has approved Sinopharm
But:
- Data behind approvals are not yet transparent
- No Indian or Chinese product is WHO pre-qualified yet
- No clarity on costs and how they will be priced for other LMICs
- Timeline for access via COVAX is unclear
- These countries might ban exports to ensure local supply
- Data behind approvals are not yet transparent
- No Indian or Chinese product is WHO pre-qualified yet
- No clarity on costs and how they will be priced for other LMICs
- Timeline for access via COVAX is unclear
- These countries might ban exports to ensure local supply
This report suggests India will not allow the export of the Serum Institute (Oxford -AstraZeneca) vaccine for several months https://apnews.com/article/ap-top-news-global-trade-immunizations-india-coronavirus-pandemic-c0c881c0f07166e8fd494e078171a7cc

 Read on Twitter
Read on Twitter