1/
[thread re sickle+thal wonderdata by @CRISPRTX]
Marking in product ~80% (SCD), 78% (TDT) = 100% edited cells - huge win for the CMC team and bodes VERY well for future of any ex vivo edited HSPC approach. Marking in marrow at 12 mo - 80% and 64%. This is MAGNIFICENT!
[thread re sickle+thal wonderdata by @CRISPRTX]
Marking in product ~80% (SCD), 78% (TDT) = 100% edited cells - huge win for the CMC team and bodes VERY well for future of any ex vivo edited HSPC approach. Marking in marrow at 12 mo - 80% and 64%. This is MAGNIFICENT!
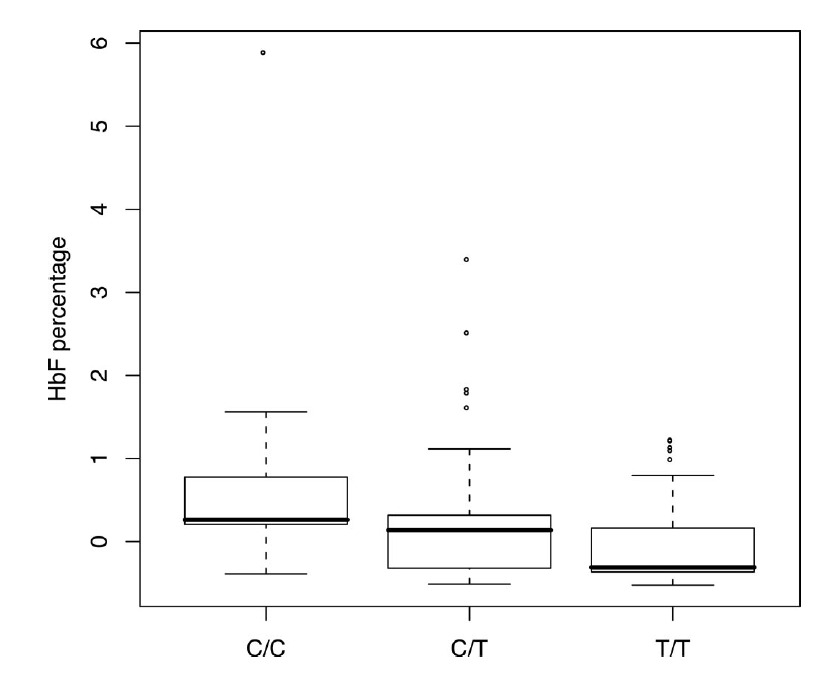
2/ 53% and 100% HbF @ 12 months - a success beyond anyone's wildest dreams. This proves that editing can make, in the clinic, a stronger allele of a GWAS hit than found in Nature (below is what the latter "does"). Paves the way for other editing trials on dialing up GWAS hits.
3/ The above adds up to a TREMENDOUS win for the patients and for science: @danielevanbauer Stu Orkin @SangamoTx @CRISPRTX.
See v nice review by Dan and Stu or one by yours truly.
https://pubmed.ncbi.nlm.nih.gov/30355263/ https://pubmed.ncbi.nlm.nih.gov/29879620/
See v nice review by Dan and Stu or one by yours truly.
https://pubmed.ncbi.nlm.nih.gov/30355263/ https://pubmed.ncbi.nlm.nih.gov/29879620/
4/ Issues to be mindful of: on the editing side, to my eye, and solely based on the data provided, no red flags. On the CMC side, note a subject - SCD-121-002 - for whom enough cells could not be obtained.
Bottom line: both subjects are clinically well = YAAY!!!
Bottom line: both subjects are clinically well = YAAY!!!
5/ This creates further HUGE motivation to develop editing for sickle/thal to be scalable, more robust, and equitable. A comprehensive editing-based cure in the next decade is realistic and a key goal for a number of groups, incl @igisci @StanfordMed @editasmed @intelliatweets

 Read on Twitter
Read on Twitter