I’m happy to share my latest paper in @J_Cell_Sci from my grad work in Karl Lechtreck's lab. Many thanks to my co-first author Aaron Harris (former lab mate),Tumo Kubo, & George Witman! Now for some context...(1/9) https://jcs.biologists.org/content/early/2020/08/13/jcs.249805
In 2015 via live-cell imaging, I found GFP-a-tubulin enters Chlamydomonas cilia by IFT & diffusion, is an IFT cargo, & incorporates into microtubules. IFT of GFP-a-tubulin is ↑↑ ~10x during ciliary growth, as is soluble tubulin. PMID:25583998 (2/9)
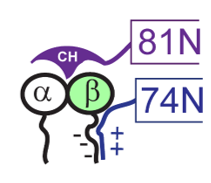
Elegant work from @esben_lorentzen described the IFT81/74 tubulin-binding module. IFT81 N-term CH domain binds a/b interface for specificity, while IFT74 N-term basic residues stabilize the electrostatic interaction w/ acidic b-tub C-term (called E-hook). PMID:23990561 (3/9)
Tumo Kubo (George Witman lab) tested this in vivo by perturbing the tubulin binding module in respective IFT mutants. Individually, IFT74 & 81 N-term mutants make full length cilia (at ↓ rates) with ↓ IFT of GFP-a-tubulin. Double mutants formed stubby cilia.PMID:27068536 (4/9)
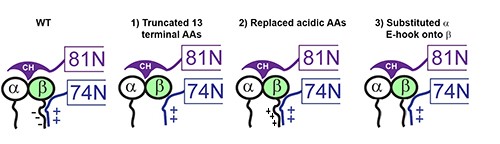
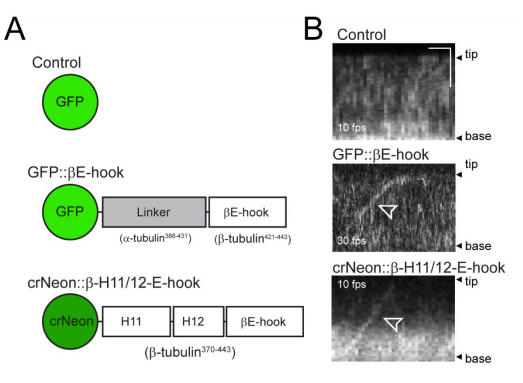
(to present paper) So, what happens if we express GFP-b-tub (~8% of total tubulin) with mutated E-hook in WT Chlamy? I made 3 constructs of GFP-b-tub: 1) truncated 13 terminal AAs 2) replaced acidic AAs to disrupt charge-based interactions 3) subbed the a-tub C-term. (5/9)
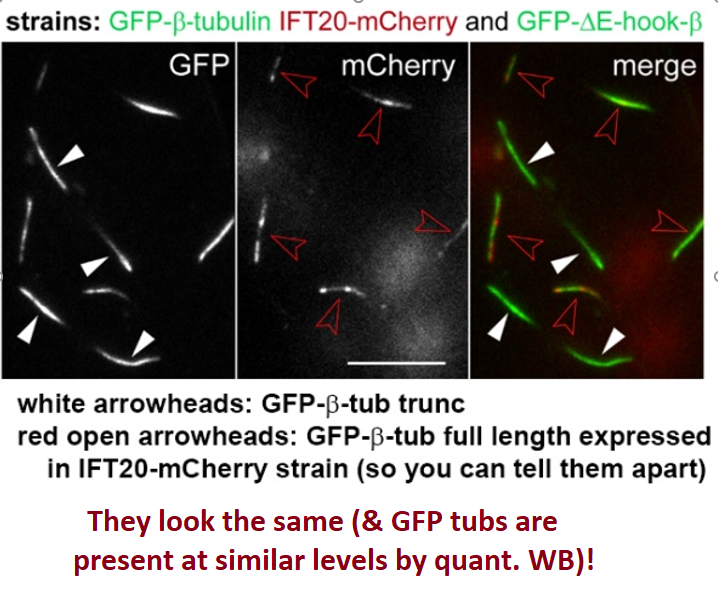
The ciliary growth rates, length, motility, IFT velocity of GFP-b-tub were normal. However, IFT frequency of the mutant tubulins were all reduced (most affected was trunc E-hook @ 90% ↓). Although largely not transported, it incorporated into axoneme at normal levels! (6/9)
This was most unexpected! We expressed trunc a & b-tub constructs in Tumo’s Chlamy strains with perturbed IFT tubulin binding module and found the a-tub E-hook is expendable for IFT, while b-tub E-hook is crucial for high frequency transport. (7/9)
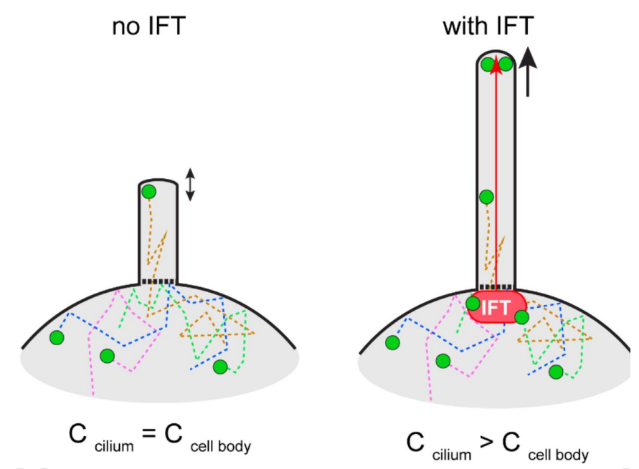
Via TIRF microscopy @ 50 fps, we observed GFP-b-tub ciliary influx by diffusion and found ~100 particles/minute enter by diffusion vs ~20 particles as an IFT cargo during regeneration. Some diffusing GFP-b-tubulin particles were immobilized at tip (=incorporation). (8/9)
We propose that most tubulin enters cilia by diffusion and can incorporate into axonemal microtubules. However, IFT of tubulin is required to exceed the critical concentration for efficient & rapid microtubule polymerization at the plus-ends at the ciliary tip. (9/9)
*Tomo Kubo*!!

 Read on Twitter
Read on Twitter