Happy to announce that our new paper that developed colorful system to understand immune evasion by transmissible cancers has been published in @ScienceAdvances https://advances.sciencemag.org/content/6/27/eaba5031
Here’s the story behind this study, how we did it, and what we found...
Here’s the story behind this study, how we did it, and what we found...
Metastatic cancer affects most mammals, but the cancer incidence can vary widely across species. Around 40% of humans and Tasmanian devils (Sarcophilus harrisii) develop cancer in their lifetime, but few cancer and immunology studies have been done in most species. @sdzglobal
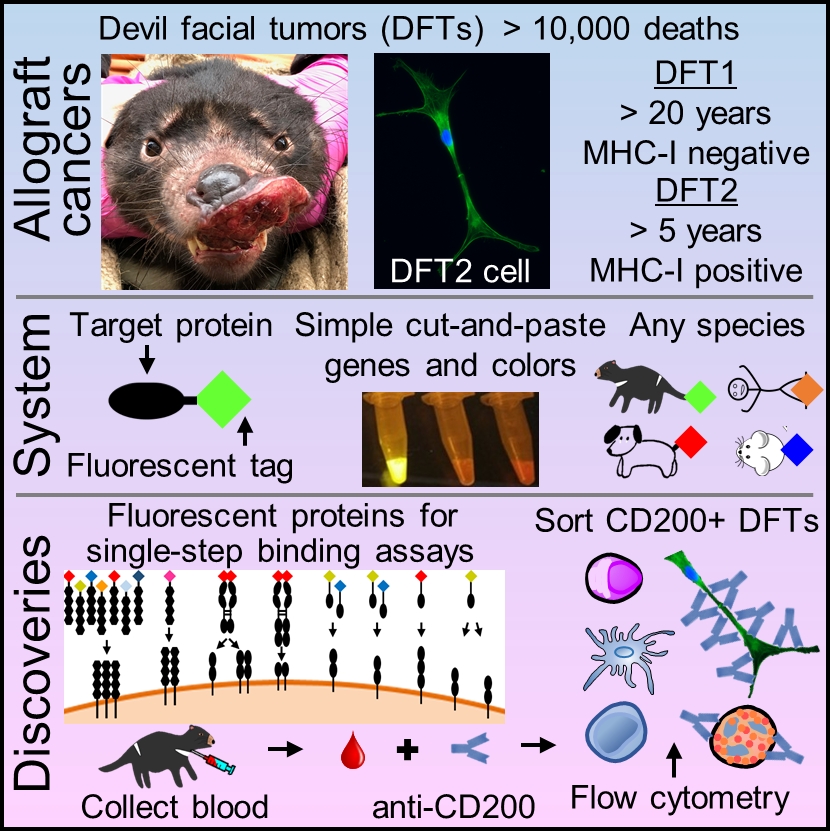
In addition to "normal cancer", Tasmanian devils are afflicted by two independent #TransmissibleCancers (aka contagious cancers) that are nearly always fatal and have led to a 77% decline in wild devil populations. Photo credit @CamilaEspejo19
The transmissible devil facial tumors (DFT1 & DFT2) are simultaneously a cancer, infectious disease, and allograft. The genetic mismatch between the host devil and the tumor cells should cause the immune system to reject the cancer...
...but only a few tumor regressions have been documented in thousands of cases of disease. This suggests the cancers are good at evading the immune system.
Immune checkpoint immunotherapy has transformed human oncology, but it potential for most non-human animals remains unknown and few tools exist to study the immune system of species other than mice and humans. https://www.cancerresearch.org/scientists/immuno-oncology-landscape/fda-approval-timeline-of-active-immunotherapies
We have previously shown that the key immune checkpoint protein PD-L1 is upregulated on DFT cells in response to interferon-gamma and developed monoclonal antibodies that can block PD-1 from interacting with PD-L1. @pseudokinase https://www.frontiersin.org/articles/10.3389/fimmu.2016.00581/full
Further comparative analysis of immune checkpoint sequences across species suggested that patterns in other checkpoint expression and binding interactions might be conserved across species, but no reagents exist for testing these. @nickomatics https://www.frontiersin.org/articles/10.3389/fimmu.2017.00513/full
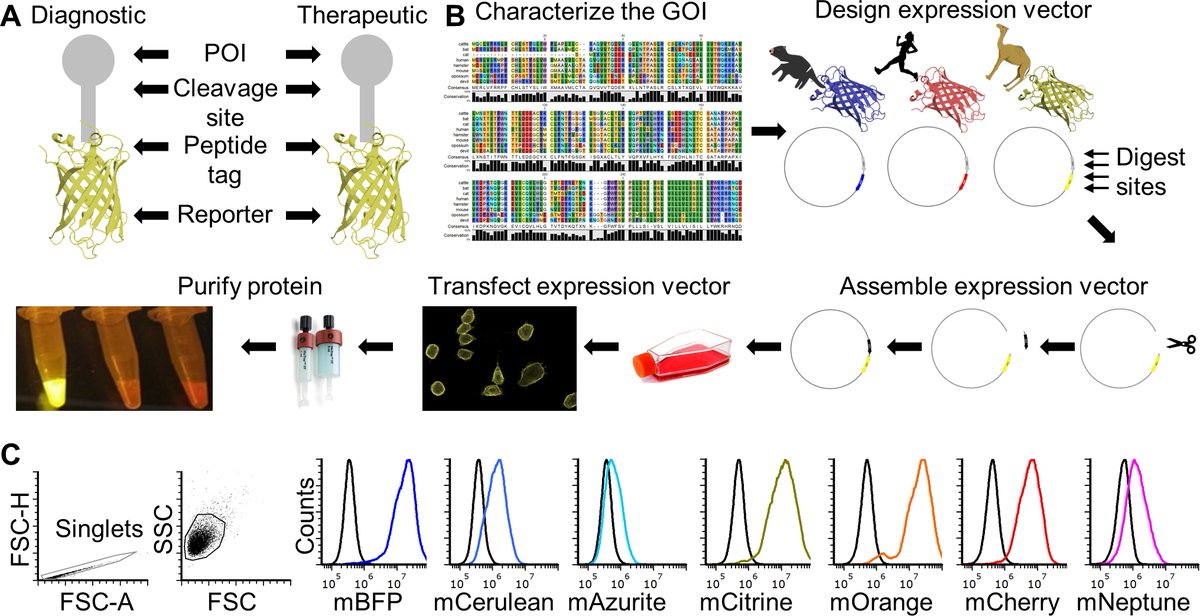
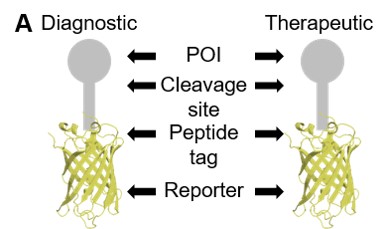
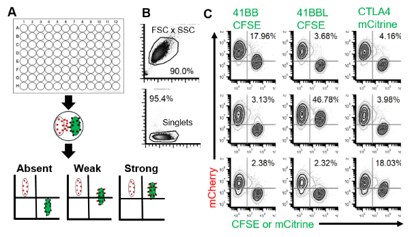
To solve the paucity of reagents available for Tasmanian devils and address ongoing limitations for nontraditional study species, we developed a Fluorescent Adaptable Simple Theranostic (FAST) protein system. @prlennard @Amanda_Patchett
The FAST proteins system uses all-in-one Sleeping Beauty expression vectors that fuses immune checkpoints to fluorescent reported protein for use as a diagnostic or therapeutic (theranostic). The #fluorescent protein can be cleaved off in downstream testing if required.
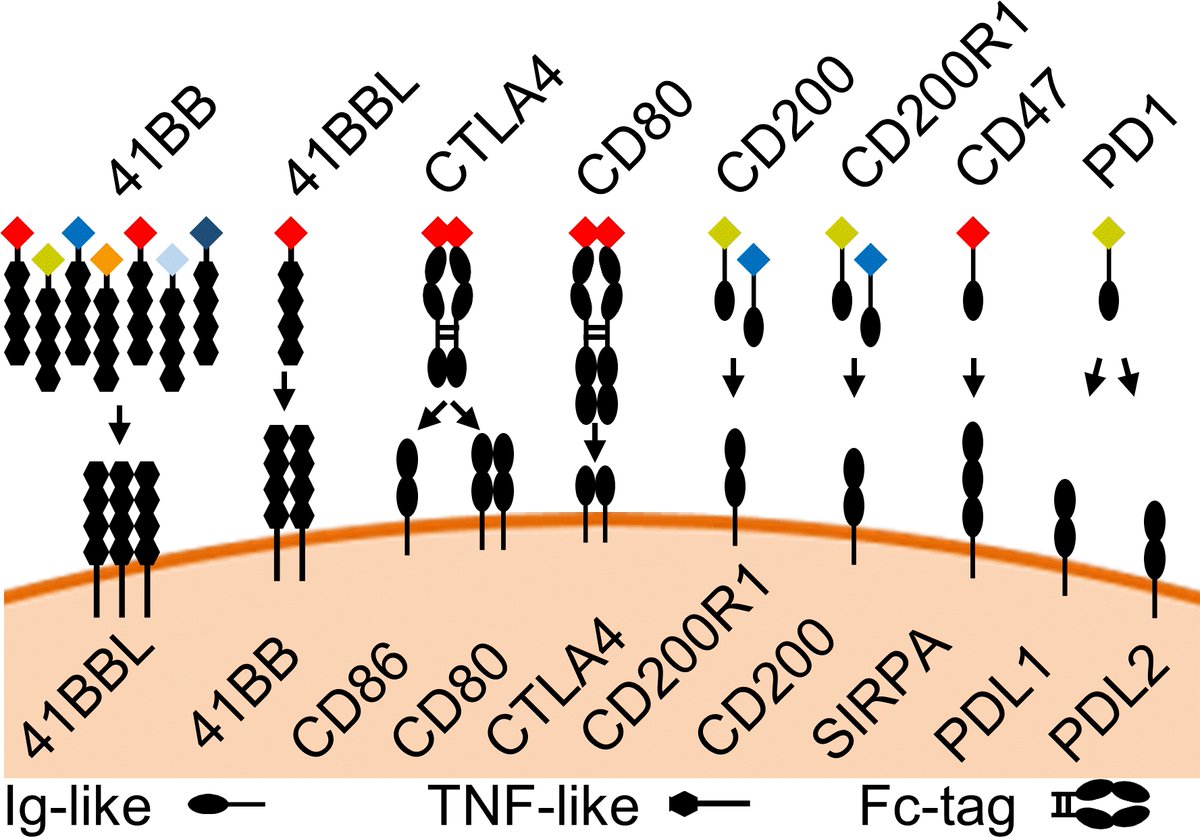
Transfection of the FAST vectors into mammalian cells results in soluble fluorescent fusion proteins that can be tested in single-step assays directly from cell culture supernatant. We confirmed seven receptor-ligand interactions among 12 checkpoint proteins.
Because the FAST proteins are secreted into the cell culture supernatant, then can be used in live coculture assays to document receptor-ligand interactions in vitro.
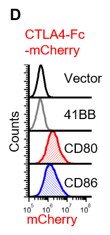
Fc-tags can be added for additional flexibility and to make dimeric proteins for increased avidity. Here we show CTLA4 fused to mCherry and binding to CD80 and CD86. @been_ong
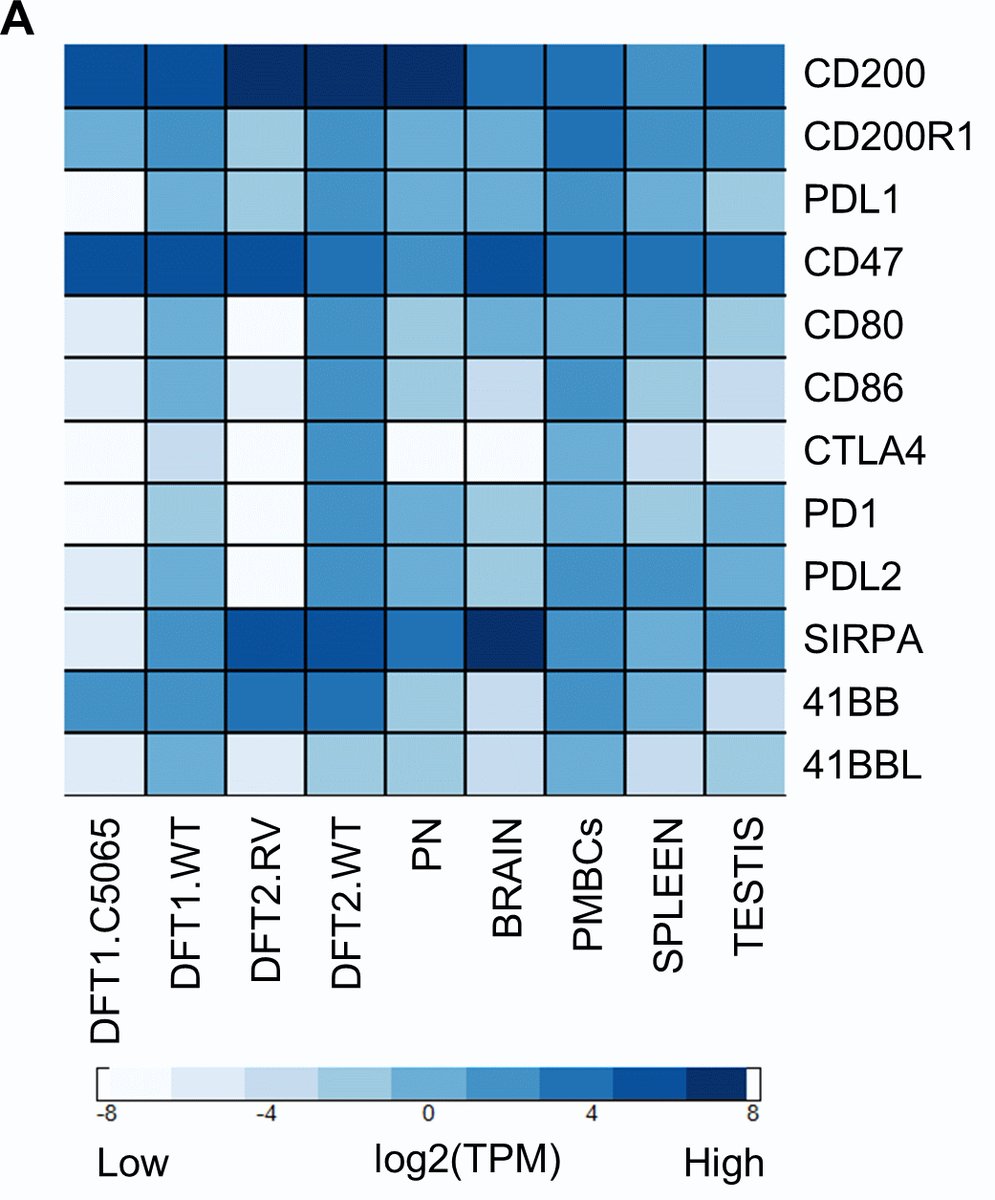
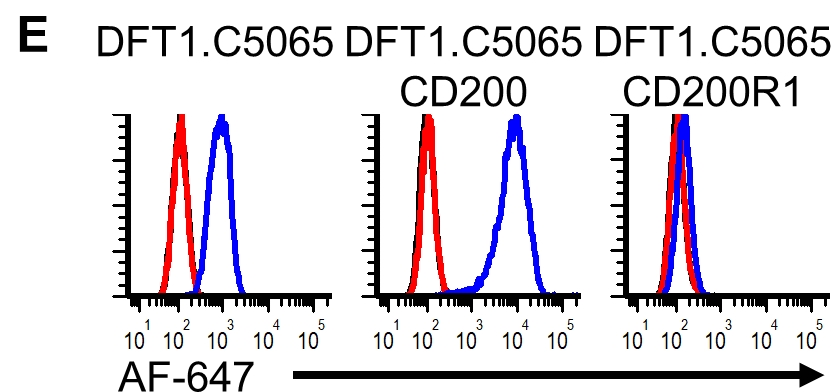
RNA-sequencing results suggested that the #CD200 immune checkpoint protein is highly expressed in DFT cells. @Amanda_Patchett
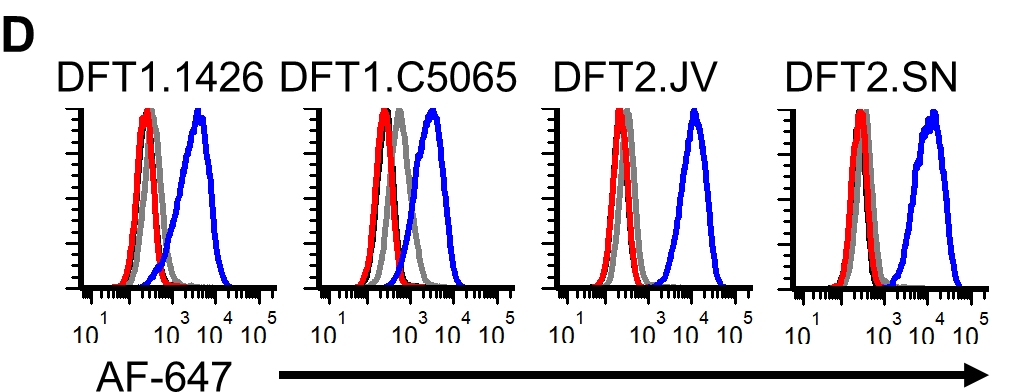
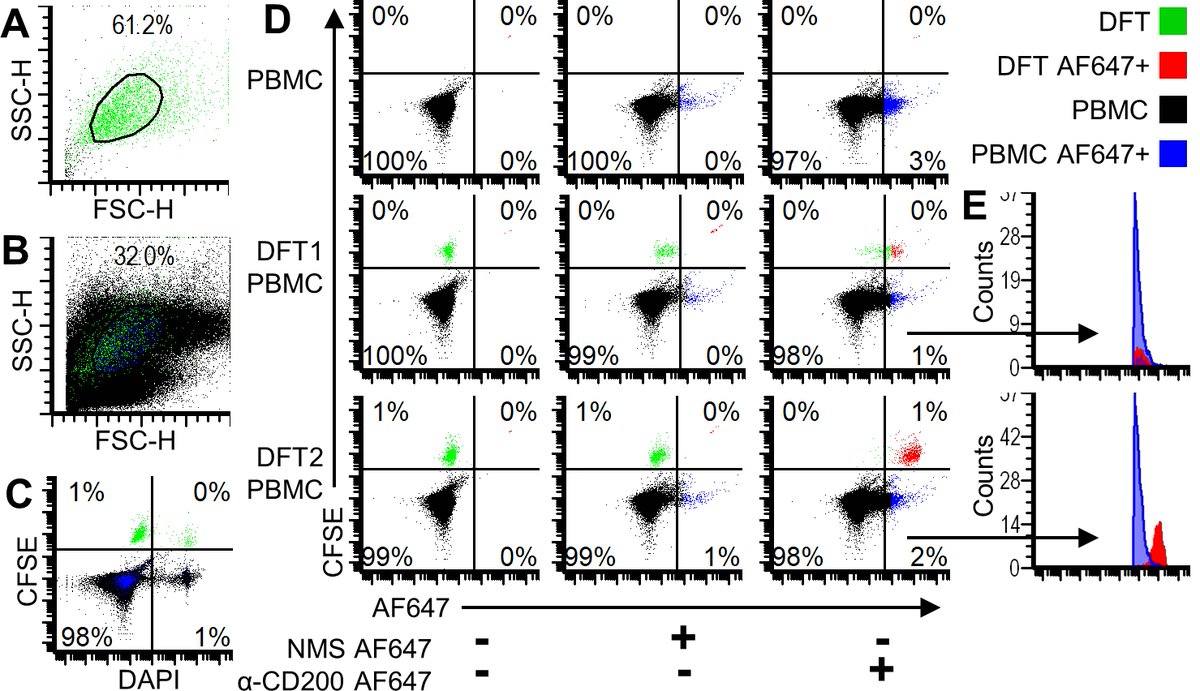
We cleaved the fluorescent reporter protein from our CD200 #FASTproteins and used it to make polyclonal anti-CD200 serum. We used the polyclonal antibodies to confirm that CD200 is highly expressed on DFT cells, particularly on DFT2 cells.
We then spiked 10,000 DFT cells into about 1 million blood cells and were able to identify the DFT cells based on CD200 expression. This opens the door to identifying circulating metastatic tumor cells in devil blood from the field.
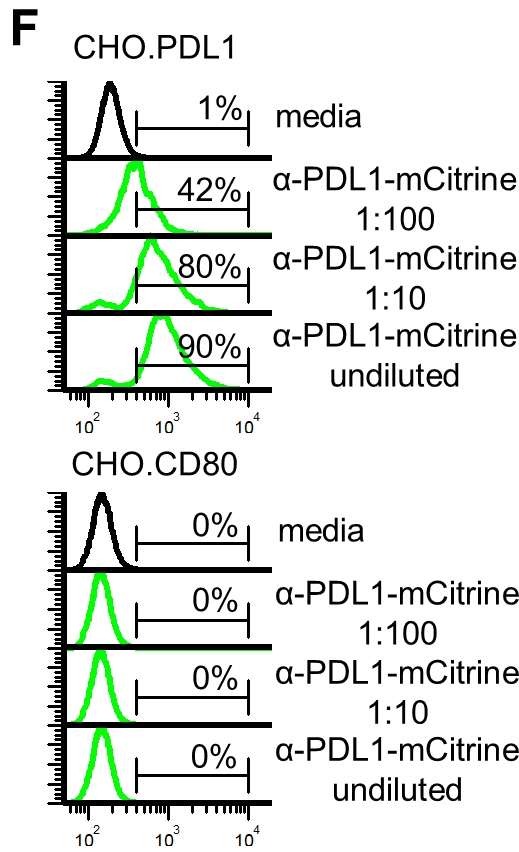
We further demonstrate the FAST system's versatility by replicating an anti-human PD-L1 nanobody that is fused to mCitrine that was used directly from cell culture supernatant to stain human PD-L1. @deluca_@alana
The plug-and-play FAST vectors can be rapidly modified for other species and to incorporate different colored fluorescent proteins.
We have a step-by-step, peer reviewed protocol for the entire FAST protein process that will be available from @bioprotocolbyte in the near future.
https://doi.org/10.21769/BioProtoc.3696
https://doi.org/10.21769/BioProtoc.3696
Our new FAST protein tools will support development of an oral bait vaccine approach to combat devil facial tumors on a landscape scale in the wild with collaborators @USDA_APHIS @mlbaker4 @AnimalSmarts @spunky_possum @SciPubTas @WACImmuno https://www.tandfonline.com/doi/abs/10.1080/14760584.2020.1711058
For the non-technical version of the story see our associated article @ConversationEDU https://theconversation.com/we-developed-tools-to-study-cancer-in-tasmanian-devils-they-could-help-fight-disease-in-humans-137710
Thank you to all of our supporters
@UTAS_ @ResearchMenzies @UTAS_MPH @UTAS_MedSci @Sciences_UTAS #DPIPWE @Emily_Flies @kayholekamp https://www.facebook.com/SaveTheTasmanianDevilAppeal/
@UTAS_ @ResearchMenzies @UTAS_MPH @UTAS_MedSci @Sciences_UTAS #DPIPWE @Emily_Flies @kayholekamp https://www.facebook.com/SaveTheTasmanianDevilAppeal/

 Read on Twitter
Read on Twitter